Frequently Accessed Pages
Effort Commitments and Payroll Certification
RAMP - Frequently Asked Questions (FAQ)
No-Cost Extension Request Procedures
Extramural Support Policies and Procedures
Research Education Development (RED)
Award Statistics FY25
Total Awards
$2092M
Federal Awards
$1007M
Non-Federal Awards
$1085M
Research Expenditures
5th (FY24)
Award Acceptance and Set-up
Page Updated: February 17, 2026
The Office of Research and Sponsored Programs (RSP) is the only office legally authorized to accept awards and agreements for sponsored programs on behalf of researchers at the University of Wisconsin-Madison. Any document requiring an official signature must be signed by an authorized representative located in RSP. Accepting awards is the function carried out by the Awards and Contracts teams in RSP. Regardless of which team in RSP handles the acceptance of an award, all awards are set-up by the RSP Awards Team. In order for a research team (PI, grant administrator, researchers) to begin spending on a sponsored award, certain steps must be taken to set up the account or authorize pre-award spending. Intended for administrators, this section covers the items needed and actions taken for sponsored project account set-up.
Award Set-up Service Expectations
The Award Acceptance & Set-up team generally operates on a first-in, first-out basis for setting up new awards and processing modifications. However, priority may be given to awards that require immediate attention to meet deadlines, like the monthly payroll calculation. It's the goal of the Award Acceptance & Set-up team to have awards set-up in as little time as possible, but also in the most compliant manner.
Award set-up will be delayed for overdue compliance items, many of which are explained in the following sections. The set-up of an award may also be delayed in times of peak activity or with unexpected absences
If you have questions regarding the status of an award, please contact the specialist assigned to the RAMP record or anyone on the RSP Award Acceptance & Set-up team.
What compliance items are reviewed before award set-up?
Effort Commitment Tracking
What is Effort and why do we need to track it?
An effort commitment is the amount of effort (time/labor) you propose in a grant proposal or other project application, and that the sponsor accepts - regardless of whether you request salary support for the effort. Commitments are specific, quantified, and can be expressed in months or as a percentage of your work over a given project period. A commitment is an obligation of effort (time/labor) that the University must fulfill.
What do I need to do?
Complete the Effort page in the RAMP Award SmartForm. Commitments are recognized, and effort must be tracked for the principal investigator/project director and key personnel listed in the Notice of Award. Update all allocation periods as appropriate. If the key personnel have changed at the time of award, update this page to reflect the current key personnel. Changes made after award set up, are not made in RAMP, but rather through the Effort Award Modification Request (AMR) type in RAMP (contact the RSP Effort Coordinator for assistance).The Effort page shows effort details for the first budget period by default, and the other periods are collapsed. Select the plus icon (+) at the far left to expand a period and the minus icon (-) to collapse a period. Enter effort in Calendar, Academic, or Summer Months for each budget allocation period at a minimum for anyone who is listed and identified in the Notice of Award as having these roles: PI, Co-PI, Key Personnel. Effort should be entered to match the Notice of Award, or if not listed in the NOA, effort should be entered based on the sponsor approved budget/proposal. Include effort for all known out-years. Enter the effort committed for each budget period in the “Months Effort” column. Enter the months requested in salary for each budget period in the “Months Requested” column. Please consider:
- Pay close attention to the terms and conditions of your award, as some awards specifically state additional names of key personnel who require commitment tracking. If such additional names are stated or referenced to, effort commitments will need to be captured.
- If the award documents are silent (do not mention anyone), the PI should still devote the minimum effort to carry out the essential scientific, administrative, and fiscal responsibilities needed for the research. This is the only effort commitment that will be tracked is the PI's commitment.
What about Cost Share?
Cost shared effort is any work on a sponsored project for which the University, rather than the sponsor, provides salary support.
Cost share is captured in the RAMP Award record SmartForm when the "Months Effort" are higher than "Months Requested" for a Person in a Budget Period. When the effort person months does not equal the requested months, the "Cost Share Months" column with auto-populate. Enter a zero (0) in the Months requested column to indicate that effort in a budget period is being cost-shared.
Guidelines for Inputting Effort
In the budget and budget justification, commitments will typically be listed in either:
- Calendar months
- Academic months
- Summer months
- Or a combination of academic and summer months
- Use Split Appointment checkbox to open the academic and summer month fields.
- For unique situations where key personnel need flexibility due to balancing many projects, specific date ranges can be given to help those projects fit into their schedules. (for example, a PI could front-load more effort into a 3 month period, and then commit less time during the remaining 9 months of the year, so long as the overall commitment is upheld over the project period). For these unique situations, please consult with your RSP Awards Setup contact.
Consideration for Specific Types of Grants
Individual schools and colleges may choose to require their own PI minimum effort level on nonfederal projects. Enforcement of such a minimum is at the discretion of each school/college at the time of division proposal review. PIs should consult with the Department Administrator and/or Dean's Office for further information on school/college-specific requirements.
- Federal Projects
- UW-Madison no longer requires a mandatory minimum 1% effort for the PI on federal and federal flow-through projects.. PIs are only required to have effort on a project when it is required by the sponsor. When no salary is requested from the sponsor, no cost share effort is necessary unless mandated by sponsor policy or a notice of award.
- It is recommended that PIs propose and devote a minimum effort to all sponsored projects to carry out the essential scientific, administrative, and fiscal responsibilities needed for the research.
- Nonfederal Projects
- While 1% PI effort is no longer required by UW-Madison policy for nonfederal awards, it is recommended that PI's propose and devote a minimum effort to all sponsored projects to carry out the essential scientific, administrative, and fiscal responsibilities needed for the research.
- Fellowships
- No effort needs to be entered for the PI or Fellow. The Effort section will have no key personnel listed.
- Supplements
- If new key personnel are being added specifically for the supplement, their effort should be input onto the supplemental project. However, the definition of a key person remains the same for supplements as primary awards.
- Diversity supplements do not require that the diversity candidate/recipient be listed as key personnel.
- Clinical Trials
- Clinical trials are a unique award type and need to be treated differently in terms of effort commitments. Generally, if the trial is supported by non-federal funds an effort commitment is not required. If the trial will be funded from federal funding a minimum commitment may be required for the PI.
- Equipment Grants
- Any awards that are solely for the purchase of equipment are exempt from the minimum effort requirements. This is any award for which 100% of the funding will be dedicated towards the purchase of a specific piece of equipment.
- Work Study
- These awards are similar to fellowships in terms of effort. No effort needs to be entered for the PI or student(s).
- Financial Aid
- No effort needs to be entered for the PI.
Overcommitments
Commitments can never total more than 100%. If a key person is over 100% when adding the new commitment to their existing workload, adjustments will need to be made, and the effort commitment on one or more projects will need to be reduced. You should work with your Effort Coordinator and key person to determine what projects need to be adjusted, and if the reduction needs sponsor approval first.
Effort Tasks
- Project-based payroll certification is UW-Madison's means of assuring compensation compliance on federally sponsored projects. Payroll certification is an after-the-fact review and attestation that the payroll charged to a federally sponsored project is reasonable in relation to the work performed. Payroll must be certified by the project's PI or their approved designee. Payroll certification requires assurance that payroll charged to a federal sponsor is reasonable in relation to work performed.
- If a PI has not completed their payroll certification tasks within the 90-day certification period, the award cannot be set up until the tasks are completed.
Financial Conflict of Interest
What is it?
The COI Program protects employees and the institution by identifying and preventing conflicts that might exist between an employee's personal interests and their public responsibilities to the University. Relationships with businesses and industries may result in the University's faculty and staff receiving financial gains from their roles in the industry - sponsored research, technology transfer, and entrepreneurship. To ensure that research is free from bias, the University is responsible for managing interactions that create potential conflicts of interest, by identifying outside financial interests disclosed in an ANNUAL electronically-submitted "Outside Activities Report." This applies to all key personnel and any individual engaged in federally funded research.
What considerations are necessary for set-up?
- New awards or contracts: Each of the key personnel in the budget/proposal will need to be listed inthe RAMP record General Tab, under the "Personnel" section. These individuals will be checked to ensure they've submitted their ANNUAL required "Outside Activities Report." Key personnel also need to be current with their COI Training Course, which is taken every 4 years.
- Identify if the individual is current with COI Training
- Identify if the individual has submitted an OAR in the last 30 days. If not, they will need to do so.
- Identify if the individual has reviewed and accepted a COI Management Plan (only applies if the COI Office has identified a potential conflict)
- Confirm that the sponsor has been notified of any potential conflicts identified during the process
- Continuation awards or contracts (amendments, modifications, incremental funding or time, etc.): Each of the key personnel listed in the budget/proposal will be checked to ensure they've submitted their ANNUAL required "Outside Activities Report".
- The above guidelines also apply to new and continuing subawards.
-
For UW System, please follow this process for System Admin personnel:
- For WI Act 36, Board of Regents Policy 13-6, and COI compliance checks, contact Diann Sypula in the UW Service Center.
- Include the following in your email:
- Subject line: System OAR Compliance Check
- Body: Full name of the individual needing the OAR check and the deadline for when the information is needed.
**Furthermore, if the specific awarding agency is a Public Health Service (PHS) agency, or another non-PHS agency that elects to use PHS regulations, additional COI activities are required for new awards. A member of the RSP award team will submit the RAMP Record to the FAST system (FCOI Award Setup Tool). If the key personnel have not submitted an updated OAR within 30 days prior to receiving an award notice, they will need to do so. Even though not all of these individuals may require effort commitment tracking, EVERY key person needs to be reviewed in the FAST system on PHS awards. The purpose of the FAST system is to:
If any potential conflicts are identified through this process, RSP reports them to the sponsor with assurance that a Management Plan is in place. This is done when RSP either submits the reports via eRA Commons, or sends an email with the FCOI information to the designated sponsor contact.
**Note: If a new key person is being added in the middle of an award funded by a PHS agency (or PHS-Reg following agency), they will need to submit an updated OAR and be reviewed in the FAST system.
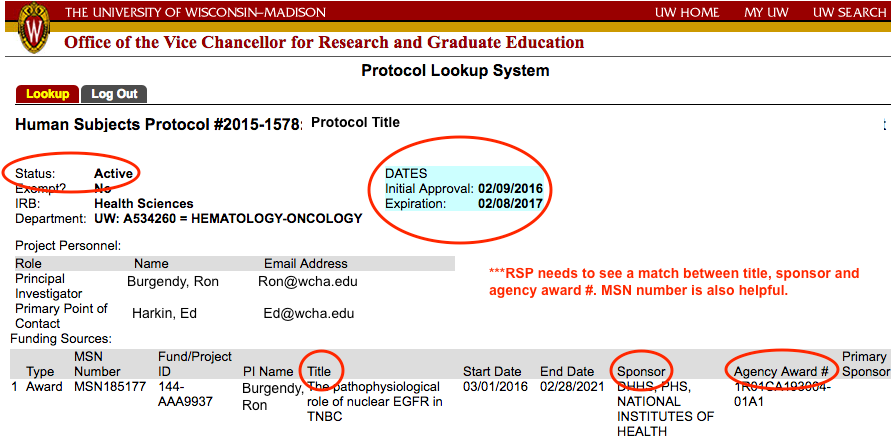
Human Subjects Protocol
What is it?
All sponsored projects with a 'yes' on the compliance question #2 located on the general tab of the RAMP record will require an approved Institutional Review Board (IRB) protocol in order to setup an award.
The Human Research Protection Program (HRPP) provides oversight for all research activities involving human participants at the University of Wisconsin-Madison. The HRPP is not an office, but rather a collective effort of all who participate in the conduct, review, approval and facilitation of human subjects research at UW-Madison. UW-Madison has two IRB committees that can review research conducted by UW investigators. UW investigators may also use external IRBs to review their projects when they are involved with collaborative research.
What do I need to do?
To complete an award set-up that includes the use of human subjects RSP will need a protocol linked to the funding proposal and displayed in RAMP. IRB protocol and funding links are initiated in ARROW by selecting the appropriate funding proposals in your IRB application. The IRB protocol information (including current status) will then automatically be displayed in the RAMP record within 24 hours. Note, funding added during a change of protocol will not be displayed until the change has been approved.
To obtain IRB protocol approval the PI will need to submit an application to the appropriate IRB office via ARROW. ARROW is the online system for preparing, submitting, reviewing, and managing applications for research oversight (protocol) at the University of Wisconsin-Madison. To find out more about starting an application in the ARROW system, click here. If you have questions, you are encouraged to contact the IRB office for additional information.
Protocol Development Activities (PDA)
What is it?
There may be times when an award has been received but the IRB protocol has not been approved yet do to the human subjects research not being conducted until later in the study. RSP will still require approval from the IRB to set-up the award. However full IRB approval may not be necessary and a PDA may be issued in place of full approval.
The Protocol Development Activities process is to facilitate the submission of grant application to the IRB for review when 1) research activities involving human subjects are planned for the future but have not been finalized; 2) when an agency or organization requires IRB approval of the concept as part of the application for funding.
What do I need to do?
In order to obtain approval to perform research activities involving human subjects, a separate Initial Review Application or Application for Exemption from IRB review will be required. For more guidance, please see the IRB PDA guidance web page.
RSP will use PLuS to confirm that a PDA has been approved for a specific project. If PLuS has not yet been updated, RSP will also accept signed documentation from the IRB stating the PDA has been approved for a specific project. When you receive a PDA please attach a copy to the RAMP record.
Ceded IRB Review
What is it?
UW-Madison may approve the use of an eternal IRB to review a human subject protocol. The external IRB may be at another institution or it may be an independent or commercial IRB. When using an external IRB, UW-Madison cedes IRB review to that IRB via a Reliance Agreement. Multi-site and collaborative research studies often use a single IRB. When that single IRB is external to UW-MAdison, Uw-Madison (and other institutions involved in the study) will rely on the single IRB. The UW-Madison IRB reliance team is responsible for ensuring appropriate reliance agreements are in place between the UW-Madison and external IRBs.
What do I need to do?
If the PI of the study would like to use another institution's IRB for review of a protocol, they should contact the IRB reliance team (irbreliance@wisc.edu) and, if appropriate, initiate a ceded IRB application in ARROW. For specifics on this process, please see the IRB's webpage for reliance agreements. Linking the funding proposal to the ARROW record for cede applications is the same as for regular IRB applications (see above). RSP will still need documentation of IRB approval from theIRB of record during award set-up.
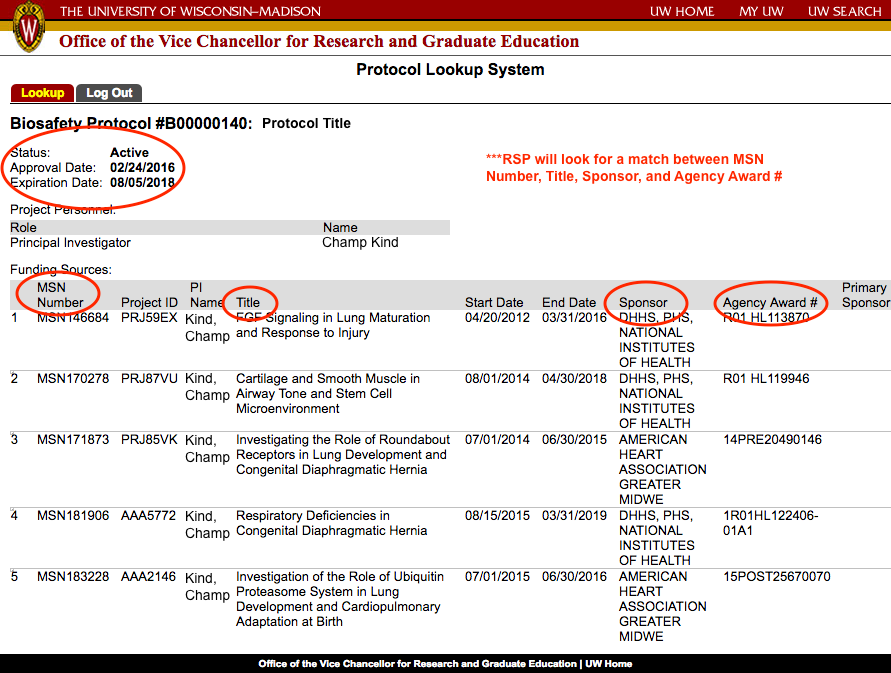
Biosafety Protocol
What is it?
All sponsored projects with a 'yes' on compliance question #1 located on the general tab of the RAMP record will need an approved Biological Safety protocol in order to setup an award. If you're unsure if a project needs an OBS protocol please see the OBS webpage for further guidance.
The UW-Madison Office of Biological Safety (OBS) assists all faculty and staff in observing safe laboratory practices for biological materials as prescribed by the Centers for Disease Control and Prevention (CDC) and the National Institutes of Health (NIH), and endeavors to assure that research is done in secure facilities in compliance with all local, state, and federal regulations. The OBS is the administrative office of the Institutional Biosafety Committee (IBC).
What do I need to do?
All sponsored projects with a 'yes' on compliance question #1 located on the general tab of the RAMP record will need an approved Biological Safety protocol in order to setup an award. RSP will not release funds until OBS approval has been documented. Detailed information regarding how to obtain an OBS protocol can be found on the OBS website along with FAQs that should help you through the process.
RSP will use PLuS to confirm that an OBS protocol has been approved for a specific project. If the protocol has not yet been updated in PLuS, RSP will also accept signed documentation from the OBS stating that a protocol has been approved for a specific project.
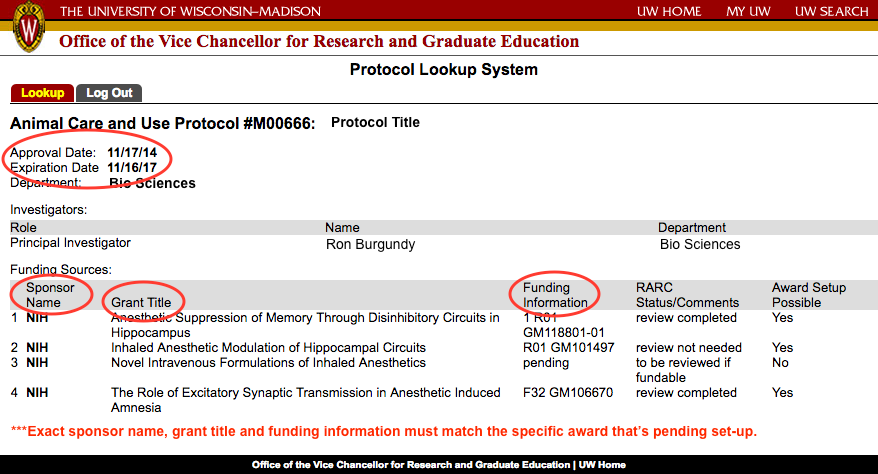
Animal Use Protocol
What is it?
RSP will review the RAMP compliance questions identify if an animal use protocol is needed. If the third compliance question "use of vertebrate animals?"" is answered as 'yes' RSP will require an approved protocol before an award can be set-up. The Institutional Animal Care and Use Committees (IACUC) are charged to oversee the use of vertebrate animals in research, teaching and education and to ensure that care and use of the animals is ethical and humane.
What do I need to do?
If the third compliance question, "use of vertebrate animals," is answered as 'yes' RSP will require an approved protocol before an award can be set-up. For more information on submitting and obtaining animal use protocol approval in ARROW, please click here.
To complete an award set-up that includes the use of animals RSP will need a protocol linked to the funding proposal and displayed in RAMP. IACUC protocol and funding links are initiated in ARROW by selecting the appropriate funding proposals in your IACUC application on the funding page. The IACUC protocol information (including current status) will then automatically be displayed in the RAMP record within 24 hours. Funding can be added to an active protocol via a non-review change which is approved upon submission.
Note: If your award is funded by the NSF or NIH and includes animal research please see the "IACUC Congruence" section.
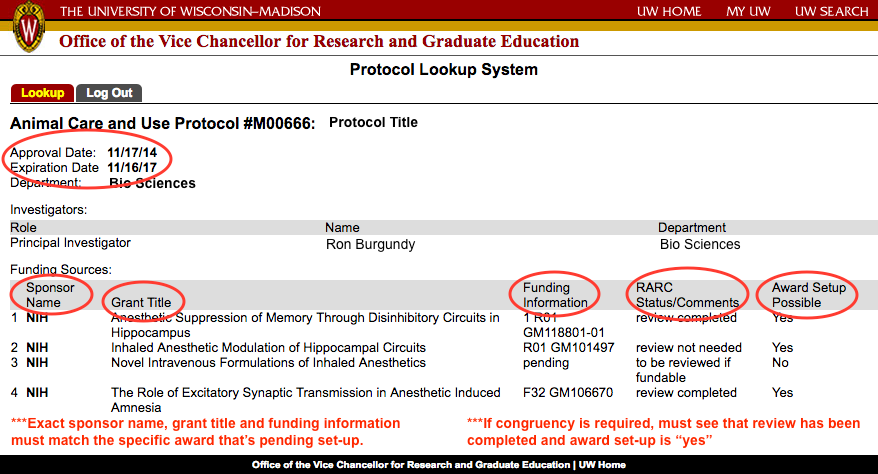
NIH & NSF IACUC Congruence
What is it?
As per the Public Health Service (PHS) Policy and NIH Grants Policy Statement, grantee organizations must establish appropriate policies and procedures to ensure the humane care and use of animals for PHS funded projects. Additionally, NIH Notice Number NOT-OD-15-139 announced an establishment of an MOU Between OLAW/OER/OD/NIH and NSF. This new agreement, effective October 1st, 2015, necessitates revisions in how UW-Madison handles NSF projects using vertebrate animals.
As part of the PHS and NSF funded award, it is an institutional responsibility to ensure that the animal activities proposed in a grant are congruent with corresponding protocols approved by the IACUC. PHS and NSF Policy requires institutions to verify that the IACUC has reviewed and approved those components of grant applications and contract proposals related to the care and use of animals. Institutions are responsible for ensuring that the information the IACUC reviews and approves is congruent with what is in the application/proposal.
What do I need to do?
All sponsored projects funded by the NSF or NIH with a 'yes' on compliance question #3 located on the general tab of the RAMP record will need a congruent, approved IACUC protocol in order to setup an award. Detailed information can be found on the RARC website regarding creating an IACUC protocol and the protocol-grant congruence review process.
PIs should ensure their active protocol includes all the animal research described in the grant, that the appropriate grant (RAMP Funding proposal) has been added to their IACUC protocol and then they can request a congruence review of the approved IACUC protocol from the study workspace in ARROW. If the grant and protocol are not congruent, RARC will notify and work with the PI to facilitate congruence and obtain IACUC approval in the most expedient manner possible. When congruence is determined, a congruence letter can be found in the IACUC study workspace in official letters tab and RAMP will be updated to indicate the protocol approval dates and congruent state.
RSP will not release funds until congruence has been documented.
Stem Cell Use Protocol
What is it?
All sponsored projects with a 'yes' on compliance question #4 located on the general tab of the RAMP record will need an approved stem cell use protocol in order to setup an award.
The Stem Cell Research Oversight (SCRO) Committee provides oversight for all research on campus or involving campus faculty or staff that involves either:
- The use of hESCs or their derivatives; or
- The introduction of hPSCs, or their derivatives, obtained from a non-embryonic source, into non-human animals at any embryonic, fetal, or postnatal stage, if an expected effect is that human cells will be integrated into the central nervous system, testes, or ovaries of the animal; or
- The storage or disposition of human embryos or gametes obtained for the purposes of stem cell research.
What do I need to do?
A Principal Investigator (PI) conducting research on either of the above must have SCRO approval in place prior to beginning research. The SCRO Committee is fully transitioned to ARROW, the online protocol application system. All initial review, change of protocol, and continuing review applications must be submitted for review through ARROW. For more information please see the SCRO webpage.
RSP will use PLuS to confirm that a protocol is approved for a specific study. If the protocol has not yet been updated in PLuS, RSP will also accept signed documentation from SCRO stating that the protocol has been approved for a specific study.
Award Terms and Conditions
What is it?
To help UW-Madison manage sponsored project funds, Workday contains a list of terms to provide supplementary information for awards. The intent is for the terms to be used as an aid in award management, although their inclusion is not a substitute for familiarity with the award document itself. Terms are selected to highlight certain conditions that govern agreements.
What is needed for award set-up?
RSP Pre-Award staff enters items into the RAMP "Terms and Conditions" tab at the time of award. When a new award is generated, the terms information flows into "Terms and Conditions" tab of the Grants/Awards module in RAMP. Campus users should contact the RSP Awards Team if an important term is missing or requires amendment.
Facilities & Administration Rates (F&A)
What is it?
Facilities & Administrative Costs are actual costs incurred by the University in support of extramural activities, but cannot be charged to a specific grant or contract. Examples include RSP services, libraries, utilities, building and equipment depreciation, and other administrative functions. They are also known as "indirect costs."
The federal government recognizes that these costs to participate in research, including expenses that are shared across all sponsored programs, cannot be allocated to one specific project. To ensure these costs are captured in the project budget, F&A is included in the budget as a percentage of the project's direct costs.
Applicable Rates
Applicable F&A rates can be found on the Rates page.
When to use an older rate vs. current rate?
Typically, the F&A rate in the proposal will remain in effect for the entire award. However, there are circumstances where an older rate is being used, but the current rate needs to be applied:
- New supplemental funds were requested and were not part of the original budget and scope of work
- Funding that wasn't previously committed at the lower rate is provided. For example, if incremental funding is provided beyond the originally proposed period of performance in the original budget and proposal, this would be considered new funding. The most current rates would then need to be built into the application, as this would fit the description of a renewal instead of a continuation.
What is needed for award set-up?
- Check to ensure that the sponsor awarded the correct F&A rate. If they did not, some negotiation may be required.
- If the incorrect rate was used at the time of proposal, the following may result:
- Re-budgeting may need to occur
- Further negotiation with the sponsor may help recover additional funds
- An F&A Waiver request may be submitted to the RSP Proposals Team. This can be done via the RAMP record:
- Send an approval using the "F&A Waiver" approval type. Please send the approval to the appropriate division.
- In addition to the approval, attach a letter to the record stating how this study and waiver request benefits the university.
- Note: Review of an F&A waiver can take 1-2 weeks as some need to be reviewed by the Vice Chancellor for Finance and Administration.
- In the projects tab, you will need to:
- Select the F&A rate type that is applicable for your award (dropdown menu)
- Select the F&A Base (dropdown menu)
- Manually enter the F&A Rate that is appropriate for your award (as a %)
- If new funding is being added to an existing award but has a different F&A rate associated with it, a separate subproject will likely need to be created. In most cases, we cannot co-mingle funds that have different applicable F&A rates.
- Procedures for Approval of Indirect Cost Exceptions at UW-Madison
Need access to the funds, but don't have an award yet?
Advance Account
What is it?
If a PI needs access to funding for a project prior to receipt of the official notice of grant award, an Advance Account may be created to allow the posting of charges to an account. The Principal Investigator's School/College/Division assumes responsibility for all charges allocated on an account in advance award status if the award is not finalized or accepted. Please see Advance Award Request Guidance for details on ADV AWD purpose, intent and use. Departments and PI's may request an advance account via RAMP. The RAMP Desk Reference walks through the process for making the request via an approval request to the division using the "Request advance award setup" approval type and then the appropriate divisional authority approving the request and notifying RSP via the Division Action page in RAMP.
Note: All compliance items will need to be addressed prior to an advance account being created including FCOI, Animal, Human, Biosafety and HESC compliance protocols and Effort Commitments.
What do I need to do?
The PI or departmental research administrator should login to RAMP and on the Approvals tab select the "request Advance Award" approval from the drop down. Note that each division may require additional justification, attachments or information in order to process this request. Please contact your Divisional Research Contact for assistance with this request. Once the division has approved the advance account request they can request the advance award set-up of RSP by selecting the "Request Advance Account of SPO" button on the division action tab in RAMP. A page will open to verify this request. Click "Process Record" to proceed. The record has been submitted to the current RSP owner for processing. Award and Project information will be reported to you via email.
What award types have special considerations?
Intergovernmental Personnel Act Assignment Agreement (IPA Agreement)
What is it?
Chapter 230.047, Wis. Stats., authorizes state agencies and the University of Wisconsin System to participate in the temporary interchange of employees between and among government agencies at the same or different levels of government and with institutions of higher education. Typical interchanges involving the University of Wisconsin-Madison are with other state agencies, federal agencies, other government agencies such as school districts, and other universities outside the University of Wisconsin System.
The purpose of an IPA is to allow an individual to remain on UW payroll and benefits while providing service to a government agency. Academic employees and departments should work directly with the funding agency and RSP to draft IPA agreements. Typically the funding agency has a standard IPA agreement that is sent to the Department/PI and a RAMP record is created after the IPA agreement is received.
Restrictions
- If a UW-Madison employee will be on assignment to a Federal agency, the arrangement must be in compliance with the Intergovernmental Personnel Act.
- An employee of the UW must be employed for at least 90 days in a career position before entering into an Intergovernmental Personnel Act agreement.
- "Career position" is defined as permanent career status receiving those employment benefits normally provided career employees of the agency, including participation in the retirement plan.
- For the William S. Middleton Memorial Veteran Hospital, an exception for an IPA for Research Associates, Research Assistants, and other training positions may be made with the written approval from the VA Hospital's HR Team.
- Employment interchanges are temporary and made for one year or less. An extension for up to one additional year may be approved. The maximum period allowed for an interchange involving the same employee is two years, including an extension.
- Employment interchange agreements are restricted to salary and fringe benefit costs.
- Reimbursement for any additional costs, such as supplies and equipment, generally may not be included as part of an employment interchange agreement.
- UW-Madison employees on assignment to another agency or institution may be eligible to receive supplemental compensation, which may be paid directly to the employee. The agency, not UW-Madison, is responsible for any tax withholding and reporting. UW-Madison employees may not receive supplemental compensation if on assignment to another State of Wisconsin agency.
- UW will not pay the travel expenses of employees incurred in connection with their work assignments at the receiving agency.
- To initiate an interchange agreement with a non-Federal agency, a Memorandum of Understanding (Appendix 2-B) should be prepared.
- Interchange agreements with a Federal agency will be initiated by that agency. Federal agencies have a specific agreement template for entering into an Intergovernmental Personnel Act agreement.
The Process
- An IPA agreement is received at the UW.
- The Department/Deans Office creates a RAMP record for the IPA, obtains all necessary approvals, completes the projects tab and submits the record to RSP.
- The record will be assigned in RSP for review of the documents to determine if IPA language is acceptable.
- RSP will negotiate if appropriate, obtain signature on the IPA and return it to the funding agency.
- RSP will review the record for project setup and send an award set up approval if appropriate.
- When the agreement is finalized RSP will set-up a 133 or 144 project for salary and fringe benefit expenditures.
Note:
- If the assignee is PI eligible, they should be listed as the contact PI on the RAMP record.
- If the assignee is not PI eligible, the assignee's faculty mentor, who is PI eligible, should be listed as the contact PI on the RAMP record.
- For IPAs with the Madison Veteran's Affairs office, all requests are processed thru the Madison VA Research HelpDesk.
What is different for set-up?
- The Award type on the RAMP General Tab should be IPA.
- The Project Type on the Projects Tab should be GM_05 Employee Interchange Agreement.
- The F&A Rate Type on the Projects Tab should be None-No F&A.
- The F&A Base Type on the Projects Tab should be No F&A (Base A).
- Total dollar amount should always be Direct Costs.
As with all other Sponsored Projects, we only capture effort for individuals identified as Key Personnel on the award. Department Personnel will make the determination and enter effort on the projects tab in RAMP only for Key Individuals.
Bench Fees
What is it?
Occasionally, visitors (e.g., students, academics, scientists) come to UW under a visiting or exchange opportunity. Such an arrangement carries with it the general purpose of educating the person and is with an educational or higher education institution. Some institutions (often international institutions) provide “bench fees” or support in the form of a check payable to UW-Madison. This is provided without application by the UW and is provided to support costs such as consumable materials, travel, and related costs associated with the visitor’s opportunity at the UW.
What's different for set-up?
- Project title as: "Bench Fees for visitor name"
- Begin date as the date of check issue unless otherwise indicated by sponsor
- End date dependent upon date of visiting arrangement ending unless otherwise indicated by sponsor
- F&A costs will not be assessed for arrangement with other non-profit institutions but will be for For-profit sponsors.
Note:
Any support deemed as bench fees which are provided by industry/for-profit-sponsors will be handled as a grant/contract (restricted) arrangement, terms dependent upon application and negotiated agreement with the sponsor.
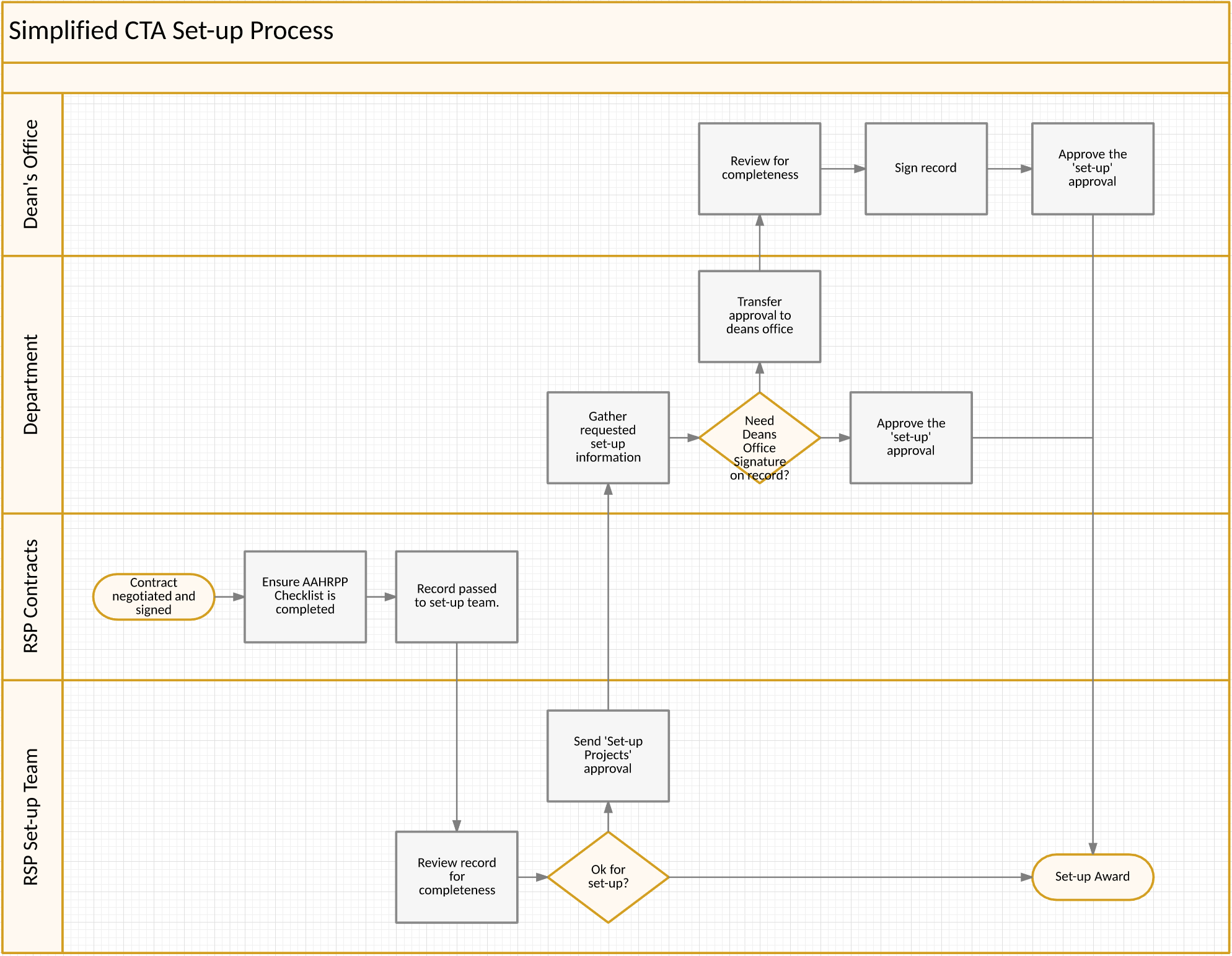
Clinical Trial Agreements (CTAs)
The Set-up Process for CTAs effective November 2017
- The RSP Contracting Group will negotiate and execute the clinical trial agreement. This will remain the same as we've historically negotiated and signed agreements.
- Upon execution of the agreement, the RSP Contracting Group will ensure the agreement and AAHRPP checklist are uploaded to RAMP. The RSP Contracting Group will forward the record to the RSP Awards Team for set-up.
- Upon receipt of the record, the Awards Team will check to see if all documentation is in the record for setup.
- If so, the Awards team will set-up the award.
- If not, a 'set-up projects' Approval will be sent to the department to complete the necessary steps (i.e. chair approvals, Ancillary Reports, IRB approvals, etc.)
- The department will complete the needed steps and respond to the 'set-up' projects approval to notify RSP they are ready for set-up.
- If division signature is needed on the record, and all other required information is attached, the department can transfer the Project Setup Approval to the Dean's Office to sign the record and respond to the approval.
- RSP will set up an account after receiving confirmation of the completed approval, and confirming that all documentation is uploaded.
Awards with Participant Support Costs
What is it?
Participant support costs are costs to support individuals who are receiving a training opportunity as part of a workshop, conference, seminar, symposium or other short-term instructional or information sharing activity funded by a sponsored award. Participant support costs are direct costs for items such as stipends or subsistence allowances, travel allowances, and registrations fees paid to or on behalf of participants or trainees (but not employees) in connection with conferences, or training projects.
A participant does not perform work or service for the project or program. The participant is not required to deliver anything or provide any service to the university in return for these support costs.
Participant support costs are typically incurred for projects that include an education or outreach component. These types of costs are most commonly included in National Science Foundation (NSF) grants, such as the Research Experience for Undergraduates (REU) or Research Experiences for Teachers (RET) program. NIH indicates that participant support costs are allowable only if specified in the Funding Opportunity Announcement.
For more information on what can and cannot be included as participant support costs, click here: Participant Support Costs Guidance
What's different for set-up?
Funds provided for participant support costs that are not spent cannot be rebudgeted for use in other categories unless prior written approval has been obtained from the sponsor.
Participant support costs are budgeted in a separate category in the application budget and must be accounted for separately. Participant support costs are excluded from the MTDC base when calculating F&A costs. RSP requires a separate project under the award to isolate participant support costs and to ensure that F&A is not applied against these costs. If possible, this additional project should be added to the Projects Tab in RAMP before the award is generated. Click the “Add Project” button on the Projects Tab to create the additional project.
- The RAMP project type to select in the Project Tab detail: GM04 (Participant Support)
- Please include in the Project Description: "Participant Support"
- F&A Rate Type: None (No F&A)
- F&A Base: No F&A
- F&A Rate: 0%
- All funds should be entered into the Direct Costs budget line
- No effort commitments are required on the Participant Support project
Award Set-up FAQs
Why does the Awards Team create new projects for some existing awards but not others?
Creation of a new project depends on the terms/requirements of the Award For example, if there is a restriction on carryover funds or if a new F&A rate is applied for the next period, a new project number will be created. A new project or award ID may also need to be created if a the sponsor issues a new subaward agreement for subsequent years of a continuing award, as opposed to issuing an amendment.
Are the approved protocols needed for an advanced account set up?
Yes. We need to verify that if the funds are released, we have the protocols in place to cover any work that is done with the funds. We will accept a PDA (protocol development activity) if IRB approval has not been obtained and human subjects work has not started. For more information on advance accounts, see above on the advance account segment.
My PI just submitted a change of protocol application in ARROW. Is a printout of the submission sufficient for set-up?
No. Proof of submission is not the same thing as an approval. RSP either needs the change to be visible in PLUS, or documentation linking the award to the protocol, such as an email from an IRB/IACUC representative, a congruence review letter, a biosafety letter, etc. Please upload any documentation to the RAMP record.
I am not receiving the Project Setup Approvals through RAMP from RSP. How do I get added to the recipient list?
This contact list is managed by your Dean's Office. Please contact them to be added.
Why is my award not set up yet? I haven't seen a project number yet.
It's possible RSP is still waiting on overdue information from the department or Sponsor. Before reaching out to RSP, please review the SPO Action Tab/notes and Approvals to ensure no overdue items are needed or we are awaiting sponsor action.
We have not received our subaward amendment for the next year of funding. Can RSP extend the project dates without an amendment from the sponsor?
No.
What happens if the sponsor reduces the initial budget period or if there is a budget reduction?
Unless otherwise notified, sponsors expect PIs (and other key personnel named in the award document) to provide the level of effort outlined in the proposal, even when the amount funded is less than requested or the initial budget period is shortened. To determine the correct baseline effort commitment, the first year’s entry in RAMP will be different than subsequent years. For example, if one committed 2 months effort and the initial budget period is reduced from 12 months to 9 months the new effort commitment percentage is 22% (= 2 months/9 months) vs. 16.7% (= 2 months/12 months). 22.22% should be entered in RAMP for the first budget period and 16.67% for all subsequent periods. This establishes the baseline commitment of 2 person months.
If at time of award, the awarded budget is reduced from the proposed budget, a PI must confirm how the project will be conducted and how funding will be allocated among budget categories, including confirmation of effort commitments and associated salary support. In doing so, PIs should carefully and deliberately manage their own (and their key personnel's) total sponsored and non-sponsored effort commitments and minimize voluntary cost sharing. A budget reduction of 25% or more from what was proposed generally indicates a project scope reduction, and a corresponding reduction in effort commitments is appropriate.
The PI is responsible for determining whether:
- The effort commitments will be reduced proportionately, and a corresponding request for approval will be sent to the sponsor. Generally, effort commitments should be reduced proportionately when the awarded budget reflects a reduction of 25% or more from the proposed budget; or
- The original effort commitments and salary support will be retained, and other budget categories can be reduced or eliminated so that voluntary cost sharing, beyond that which may have already been approved, does not occur; or
- Neither the effort commitments nor other budget categories can be reduced or eliminated. In these instances, the resulting voluntary cost sharing must be explicitly approved in accordance with the college or school’s policies on cost sharing.
For additional information, see the Effort Guidance document, specifically section 1.3.5.
Have questions?
Contact the Awards Team
For questions on any of the topics listed, please feel free to contact the specific Awards Team member who is facilitating the set-up of your award or click here for general questions/help.